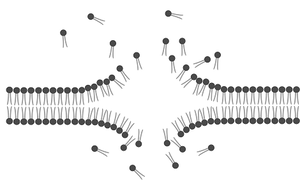
The work in our lab is highly interdisciplinary and projects are interconnected. We use biochemical and biophysical methods along with bioengineering strategies aimed at answering some of the most fundamental questions in structural biology. Using these tools, we tackle a range of research projects that include questions about intrinsic disorder in protein structure and function and their implication in disease. We are also interested in characterizing the biophysical properties of some of the materials we work with such as lipids and their ability to capture environmental pollutants.
Other projects are geared toward adapting microbes to produce high-yield expression of our proteins of interest. Toward this end we look to better understand intracellular processes that affect recombinant protein expression in heterologous organisms such as E. coli and S. cerevisiae (Baker's yeast).
We use a variety of experimental and bioinformatics tools to facilitate our projects depending on the questions we aim to answer. Students will have the opportunity to learn traditional wet lab techniques ranging from modern methods in molecular biology (cloning, PCR), clinical chemistry (ELISA) and biochemistry (electrophoresis) to biophysical techniques such as DSC to name a few. We also employ cutting edge -omics technologies (lipidomics) to analyze the lipid content in our yeast studies.
Learn more about our Research on the next page.
*This site is still under construction and undergoing changes as we continue transitioning to our new institution. Be sure to check back!
Other projects are geared toward adapting microbes to produce high-yield expression of our proteins of interest. Toward this end we look to better understand intracellular processes that affect recombinant protein expression in heterologous organisms such as E. coli and S. cerevisiae (Baker's yeast).
We use a variety of experimental and bioinformatics tools to facilitate our projects depending on the questions we aim to answer. Students will have the opportunity to learn traditional wet lab techniques ranging from modern methods in molecular biology (cloning, PCR), clinical chemistry (ELISA) and biochemistry (electrophoresis) to biophysical techniques such as DSC to name a few. We also employ cutting edge -omics technologies (lipidomics) to analyze the lipid content in our yeast studies.
Learn more about our Research on the next page.
*This site is still under construction and undergoing changes as we continue transitioning to our new institution. Be sure to check back!